SILVER SPRING, Md. — March 28, 2020 — During this COVID-19 pandemic, the U.S. Food and Drug Administration (FDA) is working around the clock to make sure that Americans have access to safe food and medical products. The agency is continuously examining the global supply chain to identify any concerns and assess the availability of the products Americans need most. We are also partnering with the Federal Emergency Management Agency (FEMA) on supply chain issues, including importation of needed medical products to support the U.S. response. Here is a status update and details on some of the latest actions we have taken:
Medical Devices
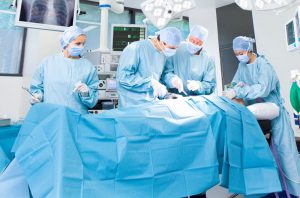
As a doctor, the situation with personal protective equipment (PPE) is very concerning. We are taking every possible action we can, but we know the situation is dire for many health care facilities. We want to share that the FDA, along with the private sector, FEMA and all of the U.S. Government, are working around the clock to address this issue as quickly as possible.
We are aware of shortages of respirators and have issued Emergency Use Authorizations (EUA) covering certain respirators to enable them to be used in health care settings. Via the EUA and the shortages mailbox, the team has directed stakeholders to CDC’s strategies for optimizing respirator supply and recommendations on the use of expired respirators.
Most recently, the FDA issued an EUA to help increase the supply of filtering facepiece respirators by authorizing the use of certain imported disposable respirators that are not National Institute for Occupational Safety and Health (NIOSH)-approved for use by health care professionals. We are continuing to work on strategies to increase the availability of these devices.
We are also working on mitigation strategies for other types of personal protective equipment including surgical masks, gowns, and gloves. We have issued information to health care providers and have provided recommended conservation strategies for these important PPE.
We are working with textile manufacturers, including clothing designers, about how we can repurpose their manufacturing product lines to put them to use to make masks. These masks can be used for surgical or other isolation procedures, as the benefits of using them outweigh the risks under current circumstances.
We are providing maximum regulatory flexibility where we can.
We are also open to importing PPE and other devices and have provided instructions to manufacturers to help us implement this necessity appropriately. The agency is taking steps to facilitate importation of PPE into the U.S. and we are ready and available to engage with importers to minimize disruptions during the importing process. This means that when products come in from overseas, their authorization via our EUA authority allows them to quickly enter the country. These products aren’t hindered from entry and we’ve already determined, among other criteria, that the known and potential benefits of these products being used in health care settings outweigh the known and potential risks.
The FDA has reached out to more than 1,000 device manufacturing sites worldwide, focusing on essential devices. The outreach thus far has focused on two main types of essential devices: those that are in high demand due to the pandemic outbreak, such as PPE and ventilators, and devices that may be prone to potential shortage if there is a supply disruption.
We further recognize the need for ventilators, ventilator accessories, and other respiratory devices may outpace the supply available to health care facilities. We have issued an immediately in effect guidance outlining a policy intended to help increase availability of ventilators and their accessories as well as other respiratory devices. In addition to the guidance, the FDA has issued an EUA that authorizes the emergency use of ventilators, anesthesia gas machines modified for use as ventilators, and positive pressure breathing devices modified for use as ventilators (collectively referred to as “ventilators”), ventilator tubing connectors, and ventilator accessories, that FDA determines meet the specified criteria for safety, performance and labeling. We anticipate this action will increase the number of patients who can have access to ventilators or similar machines for respiratory distress, which is important for critically ill COVID-19 patients
As with prior emergencies, the FDA has taken proactive steps to establish and remain in contact with medical device manufacturers and others in the supply chain, including hospitals and group purchasing organizations. The agency also encourages manufacturers and healthcare facilities to report any supply disruptions to the device shortages mailbox, deviceshortages@fda.hhs.gov. This mailbox is closely monitored and has proven to be a valuable surveillance resource to augment FDA efforts to detect and mitigate potential supply chain disruption.
Human Drugs & Biologics and Animal Drugs
We continue to work closely with manufacturers to make sure that they continue to notify the FDA, as early as possible, of a permanent discontinuance or any interruption in manufacturing that is likely to lead to a disruption in supply. This communication and the full cooperation of companies providing specific and necessary information is imperative in order for us to have an accurate understanding of the supply landscape and work to take proactive steps to mitigate shortages. To help human drug manufacturers submit timely and informative notifications, the agency is publishing a guidance for immediate implementation about the importance of these notifications, the timelines for manufacturers to follow when notifying the FDA, and the details for them to provide about the discontinuance or interruption of manufacturing. We recognize that while some supply disruptions and shortages cannot be predicted or prevented, early communication and detailed notifications from manufacturers to the agency play a significant role in decreasing their incidence, impact, and duration.
Our efforts with federal partners and pharmacy groups continue to establish mitigation strategies and prevent long-term supply shortages. At this time, where there is an acute demand for certain products and disruption in the supply chain, we are taking proactive steps to make sure that patients can access the medications that are medically appropriate and necessary. Some retail pharmacies have begun implementing policies designed to limit the overprescribing and overdispensing of certain medications in order to preserve the supply nationally. We are also aware that a number of states have taken action to limit the dispensing of certain drugs, including chloroquine and hydroxychlorquine.
Our public drug shortages lists are up-to-date with human and animal drugs and biological products that we have determined to be in shortage. These shortages are not all results of COVID-19, with many existing prior to the pandemic as results of market changes and supply challenges. We are updating these lists regularly and communicating in real-time so that patients and healthcare providers have the most current information on product shortages in the U.S.
The agency has decided it does not intend to object to registered outsourcing facilities using hydroxychloroquine. Despite this action, it is important to note that there are currently no FDA-approved therapeutics or drugs to treat, cure or prevent COVID-19; however, there are FDA-approved treatments that may help ease the symptoms of COVID-19.
Lastly, we appreciate industry’s willingness to help supply hand sanitizer to the market to meet the increasing demand for alcohol-based hand sanitizers during the COVID-19 pandemic. As part of our commitment to providing timely guidance to support continuity and response efforts during this pandemic, the agency has published guidances to provide flexibility to help meet the demand for hand sanitizer.
Blood Supply
Blood supplies have been dramatically reduced due to the implementation of social distancing and the cancellation of blood drives. Maintaining adequate levels of our nation’s blood supply is critical. People who donate blood are equivalent to those who work in critical infrastructure industries. We continue to encourage individuals who are healthy and able to donate blood. Donating is safe and takes only a little of your time. At many blood donations centers, those who are interested in donating can make an appointment to minimize the time it takes to donate blood. Contact information for local blood collection sites and for scheduling appointments can be found on FDA’s website.
Human and Animal Food
Overall, retail supply chains remain strong, and we are working with food manufacturers and grocery stores to closely monitor the human and animal food supply chain for any shortages. The agency is in close contact with industry and its trade associations, which are in touch with their members about supply chain issues.
Food production and manufacturing – for both people and animals – are dispersed throughout the U.S., and we are pleased to report there are currently no widespread disruptions reported in the supply chain. Empty grocery shelves are largely due to unprecedented demand – not a lack of capacity to produce, process and deliver. Manufacturers and retailers are working around the clock to replenish shelves.
The same is true for animal food. The FDA is monitoring the availability of foods for livestock and pets. There are no shortages, and no current disruptions in the pet and livestock food supply chain.
I am grateful to workers in the food and agriculture sector filling critical and essential roles within communities. Their ability to continue to work during periods of community restrictions, social distancing, and closure orders, among others, is crucial to community continuity and community resilience. This was reinforced by DHS in its Guidance on the Essential Critical Infrastructure Workforce: Ensuring Community and National Resilience in COVID-19.
Veterinary Medicine
Our Center for Veterinary Medicine is working with veterinary stakeholder groups to alert veterinary professionals to adopt PPE conservation strategies to make the most of their current supply of items, such as masks and gloves. We’re also encouraging the postponement of non-urgent/routine veterinary medical procedures during this pandemic. In addition, we have been coordinating with veterinary medical supply manufacturers and distributors to explore how certain veterinary medical supplies, such as sterile swabs, and devices, such as ventilators, could be redirected to address critical shortages in human health care.
Posted March 30, 2020
Source: U.S. Food and Drug Administration
 ZURICH — March 31, 2020 — A new Secretary General has been chosen by Cematex, the European Committee of Textile Machinery Manufacturers. Cornelia Buchwalder has served on an interim basis since June 2019, following the retirement of Maria Avery, and was elected to the permanent position by the Cematex Board on March 12.
ZURICH — March 31, 2020 — A new Secretary General has been chosen by Cematex, the European Committee of Textile Machinery Manufacturers. Cornelia Buchwalder has served on an interim basis since June 2019, following the retirement of Maria Avery, and was elected to the permanent position by the Cematex Board on March 12.